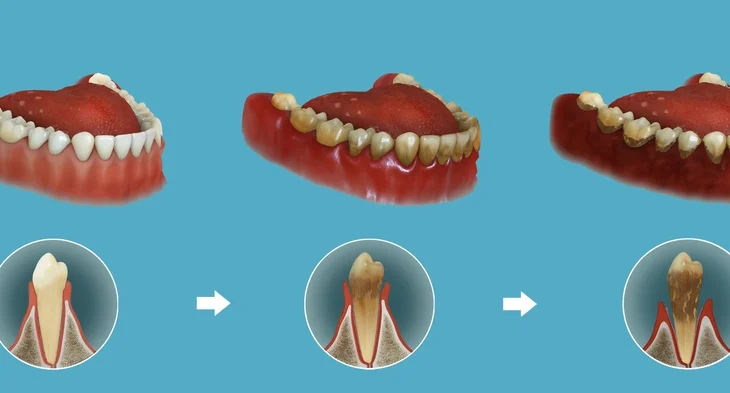
The health of the periodontium — the supporting structures of the teeth — is fundamental to oral and overall well-being. Periodontal diseases, encompassing gingivitis and periodontitis, are among the most common chronic inflammatory conditions affecting humans. While gingivitis refers to inflammation limited to the gingival tissues, periodontitis represents a more destructive process involving the loss of connective tissue attachment and alveolar bone.
Although both conditions share similar microbial origins and inflammatory pathways, they differ significantly in their clinical implications, reversibility, and management approaches. Importantly, gingivitis does not always progress to periodontitis; in many individuals, the inflammatory response remains confined to the gingival margin. Understanding the biological mechanisms and clinical features of these diseases is essential for accurate diagnosis, prevention, and effective intervention.
Table of Contents
TogglePathogenesis of Gingivitis and Periodontitis
Periodontal diseases arise from a dynamic interplay between microbial biofilms and the host’s immune-inflammatory responses. The progression from health to gingivitis and then to periodontitis represents a continuum of host-microbial interaction rather than a set of discrete entities.
The primary etiological factor is dental plaque — a structured microbial biofilm that accumulates on tooth surfaces, especially in the gingival crevice. This biofilm triggers a sequence of inflammatory and immune reactions in the gingival tissues, leading to characteristic histological and clinical changes.
The Biofilm and Host Response
The bacterial biofilm in the gingival sulcus is composed of diverse microorganisms, including Streptococcus, Actinomyces, Fusobacterium, Prevotella, and Porphyromonas gingivalis. In health, a delicate equilibrium exists between these microorganisms and the host defense mechanisms. However, poor oral hygiene, systemic factors, or immune compromise can disrupt this balance, leading to microbial dysbiosis and inflammation.
The host response — not the bacteria alone — is the primary determinant of tissue destruction. Cytokines, prostaglandins, and matrix metalloproteinases (MMPs) released by neutrophils, macrophages, and fibroblasts mediate connective tissue degradation and alveolar bone resorption.
Histopathological Stages of Gingival Inflammation
Classic studies, notably those by Page and Schroeder (1976), described four distinct but overlapping histopathological stages in the development of gingivitis and its potential progression to periodontitis:
- Initial lesion
- Early lesion
- Established lesion
- Advanced lesion
These stages represent a continuum of increasing inflammatory activity and tissue damage.
The Initial Lesion (0–4 days)
The initial lesion represents the earliest response of gingival tissues to microbial challenge. It occurs within 2–4 days of plaque accumulation.
- Vasodilation and Increased Permeability:
Localized vasodilation occurs in the gingival connective tissues adjacent to the junctional epithelium. The resultant increase in vascular permeability allows plasma proteins, including antibodies and complement components, to migrate into the tissues and sulcular fluid. - Gingival Crevicular Fluid (GCF):
There is an increased flow of GCF, which serves as a flushing mechanism, removing bacterial toxins and metabolic byproducts from the gingival sulcus. - Cellular Infiltrate:
Neutrophils (polymorphonuclear leukocytes) are the predominant immune cells infiltrating the junctional epithelium and sulcus. Small numbers of lymphocytes and macrophages may also appear. - Clinical Appearance:
Despite these microscopic changes, the gingiva appears clinically healthy. No visible redness, swelling, or bleeding is evident at this stage.
The initial lesion demonstrates that subclinical inflammation can exist even in apparently healthy gingiva, emphasizing the importance of plaque control.
The Early Lesion (4–7 days)
After about one week of continued plaque accumulation, the early lesion develops.
- Vascular Changes:
There is an increase in the number and diameter of vascular units, leading to visible erythema (redness) of the marginal gingiva. - Inflammatory Cells:
Neutrophils and lymphocytes dominate the lesion, with a small number of macrophages present. Plasma cells are few. - Connective Tissue Alterations:
Fibroblast degeneration begins, and collagen fibers, especially those near the junctional epithelium, break down. The basal cells of the junctional epithelium proliferate and form rete pegs extending into the connective tissue. - Biofilm Expansion:
Subgingival biofilm begins to develop as the junctional epithelium loses its tight contact with the enamel surface. - Clinical Features:
The gingiva becomes reddish, swollen, and bleeds on probing. At this stage, the lesion may persist without progression if adequate plaque control is established.
The Established Lesion (2–3 weeks)
The established lesion represents chronic gingivitis, which may remain stable for long periods or progress to destructive disease.
- Predominant Immune Cells:
Plasma cells now increase significantly, though lymphocytes and neutrophils remain abundant. - Gingival Crevicular Fluid Flow:
The flow of GCF continues to rise, delivering inflammatory mediators into the sulcus. - Junctional Epithelium:
The epithelium becomes increasingly permeable and may migrate slightly apically, covering part of the root surface. - Connective Tissue:
Collagen loss becomes more extensive, and fibroblast function is compromised due to persistent inflammation. - Clinical Manifestations:
Gingiva appears red, swollen, soft, and bleeds easily on gentle probing. The texture is often smooth and shiny. Despite the chronic inflammation, no attachment loss or bone loss has yet occurred.
The established lesion may persist for months or years with no progression, representing a stable inflammatory state.
The Advanced Lesion (Periodontitis)
The advanced lesion marks the transition from gingivitis to periodontitis — a state characterized by irreversible tissue destruction.
- Apical Migration of Junctional Epithelium:
The junctional epithelium migrates apically, forming a true periodontal pocket lined by pocket epithelium. - Inflammatory Cell Infiltration:
Plasma cells dominate (>50% of the infiltrate), accompanied by lymphocytes and macrophages. The inflammation extends deeply into the connective tissue and alveolar bone. - Connective Tissue and Bone Destruction:
The inflammatory mediators, particularly interleukin-1β (IL-1β), tumor necrosis factor-alpha (TNF-α), and prostaglandin E2 (PGE2), stimulate osteoclastic bone resorption and matrix breakdown. - Clinical Attachment Loss (CAL):
This stage manifests as measurable attachment loss (1–2 mm initially) and pocket formation. Early lesions exhibit shallow suprabony pockets; as disease advances, bone loss becomes vertical with infrabony pockets. - Clinical Appearance:
Gingiva appears bluish-red, spongy, and may show recession. The teeth may become mobile or drift due to loss of supporting bone.
The advanced lesion represents established periodontitis, a chronic condition that requires professional intervention.
Mechanisms of Tissue Destruction
The transition from gingivitis to periodontitis is not merely bacterial overgrowth but an imbalance between microbial challenge and host defense.
- Enzymatic Degradation:
Bacterial enzymes such as collagenases and proteases directly degrade connective tissue components. - Host-derived Mediators:
Host immune cells release cytokines (IL-1, TNF-α), chemokines, and MMPs that exacerbate tissue breakdown. - Bone Loss:
Bone resorption is mediated by activation of the RANK/RANKL/OPG pathway. Osteoclasts are stimulated by inflammatory mediators to resorb alveolar bone. - Host Susceptibility:
Genetic predisposition, smoking, diabetes, and systemic diseases influence the severity and rate of progression.
Periodontitis, therefore, represents a dysregulated host response to a persistent microbial biofilm rather than a purely infectious disease.
Clinical Features of Gingivitis
Clinically, gingivitis presents with a characteristic triad of redness, swelling, and bleeding on gentle probing. These signs are readily observed during routine dental examination.
Classical Signs and Symptoms
- Color: The gingiva changes from a coral pink to reddish or bluish-red hue due to increased vascularity.
- Contour: The marginal gingiva loses its “knife-edge” appearance and becomes rounded.
- Consistency: The tissue becomes soft and spongy due to edema.
- Surface Texture: The normal stippling disappears, giving a shiny appearance.
- Bleeding: Bleeding on probing or brushing is a hallmark sign.
- Pain: Usually absent; some patients may report mild tenderness.
- Halitosis: Bad breath can occur due to bacterial metabolites.
Probing Depth and Attachment
In gingivitis, probing depths >3 mm may occasionally be observed. However, this does not represent true attachment loss — rather, it results from gingival enlargement due to edema or hyperplasia. There is no apical migration of the junctional epithelium, and the alveolar bone remains intact.
Reversibility
Unlike periodontitis, gingivitis is completely reversible with effective plaque control and professional cleaning. If left untreated, however, chronic inflammation may eventually extend to the deeper supporting structures.
Clinical Features of Periodontitis
When the inflammatory process extends beyond the gingiva to involve the periodontal ligament and alveolar bone, periodontitis develops. It is characterized by attachment loss, bone resorption, and formation of periodontal pockets.
Chronic Periodontitis
Chronic periodontitis is the most common form seen in adults.
Clinical Features:
- Gingival inflammation and bleeding during brushing or probing.
- Pocket formation due to apical migration of the junctional epithelium.
- Tooth mobility and migration, especially in anterior teeth.
- Recession of the gingival margin, leading to root exposure.
- Halitosis and unpleasant taste.
- Tooth sensitivity to temperature changes due to exposed roots.
Pocket Formation and Bone Loss
Periodontal pockets represent a pathological deepening of the gingival sulcus. They may be:
- Suprabony pockets: where the base of the pocket is above the alveolar crest.
- Infrabony pockets: where the base is below the alveolar crest.
Radiographically, bone loss may appear horizontal or vertical, depending on the pattern of resorption.
Disease Distribution
- Localized Periodontitis: Involves less than 30% of sites in the mouth.
- Generalized Periodontitis: Involves 30% or more of sites.
The extent and severity depend on host susceptibility and local contributing factors such as plaque retention and calculus.
Disease Progression
The stage of periodontitis describes the severity and extent of tissue destruction, whereas the grade indicates the rate of progression.
- Stage I–IV: Based on the degree of clinical attachment loss and radiographic bone loss.
- Grade A–C: Reflects slow, moderate, or rapid disease progression, respectively.
Systemic Influences
Several systemic conditions can exacerbate periodontal destruction:
- Diabetes mellitus impairs neutrophil function and collagen metabolism.
- Smoking alters vascularity and immune response, masking bleeding and promoting bone loss.
- Hormonal changes, stress, and genetic predisposition can also contribute.
Classification of Periodontal Health and Disease
The 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases introduced updated criteria emphasizing clinical attachment and probing findings.
Patients with an Intact Periodontium
| Condition | Probing Attachment Loss | PPD | BOP | Radiographic Bone Loss |
|---|---|---|---|---|
| Health | No | ≤3 mm | <10% | No |
| Gingivitis | No | >3 mm | >10% | No |
Patients with a Reduced Periodontium
| Condition | Probing Attachment Loss | PPD | BOP | Radiographic Bone Loss |
|---|---|---|---|---|
| Health | Yes | ≤3 mm | <10% | Possible |
| Gingivitis | Yes | >3 mm | >10% | Possible |
This classification recognizes that patients who have undergone periodontal therapy (hence, with a reduced periodontium) can still achieve a healthy status if inflammation is controlled.
Radiographic and Clinical Correlation
Radiographs are invaluable in diagnosing and monitoring periodontitis. A DPT (Dental Panoramic Tomogram) can reveal the typical pattern of bone loss. The case example from the text (Fig. 5.2) illustrates established periodontitis in a diabetic smoker — highlighting the multifactorial nature of the disease.
Clinically, periodontal probing and measurement of clinical attachment loss (CAL) remain the gold standards for assessing disease severity and progression.
Prevention and Management
Preventive Strategies
- Mechanical plaque control: Brushing, interdental cleaning, and professional scaling.
- Chemical plaque control: Use of chlorhexidine or essential oil mouthrinses as adjuncts.
- Patient education: Emphasizing proper oral hygiene and regular dental visits.
Professional Management
- Non-surgical therapy: Scaling and root planing to remove plaque and calculus.
- Adjunctive antimicrobial therapy: Local or systemic antibiotics where indicated.
- Surgical therapy: Flap surgery, bone grafting, and regenerative procedures for advanced cases.
- Supportive periodontal therapy (SPT): Long-term maintenance is crucial to prevent recurrence.
Prognosis and Disease Progression
The course of periodontal disease varies widely between individuals and even among different sites within the same mouth. Some lesions progress linearly, while others follow a burst model — with periods of rapid destruction interspersed with inactivity.
Key prognostic factors include:
- Patient’s oral hygiene practices
- Systemic health (e.g., diabetes)
- Smoking habits
- Genetic predisposition
- Stress and immune status
Early detection and intervention are vital to prevent irreversible tissue loss.
Conclusion
Gingivitis and periodontitis represent a biologically interconnected spectrum of inflammatory diseases affecting the periodontium. Gingivitis, while reversible, serves as a warning sign of microbial challenge and potential progression. Periodontitis, on the other hand, signifies irreversible destruction of connective tissue and bone — a process governed by the complex interplay of biofilm pathogens and host immune responses.
Understanding the pathogenesis, histopathology, and clinical features of these diseases allows clinicians to implement preventive strategies, make accurate diagnoses, and tailor management plans to individual patient needs.
Periodontal health is not merely the absence of disease but a state of harmonious balance between microbial ecology and host defense — a balance that can be maintained through diligent oral hygiene, patient education, and professional care.
References
- Page, R. C., & Schroeder, H. E. (1976). Pathogenesis of inflammatory periodontal disease: A summary of current work. Laboratory Investigation, 33(3), 235–249.
- Lindhe, J., Lang, N. P., & Karring, T. (Eds.). (2015). Clinical Periodontology and Implant Dentistry (6th ed.). Wiley-Blackwell.
- Newman, M. G., Takei, H., Klokkevold, P. R., & Carranza, F. A. (2019). Carranza’s Clinical Periodontology (13th ed.). Elsevier.
- Tonetti, M. S., Greenwell, H., & Kornman, K. S. (2018). Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. Journal of Periodontology, 89(Suppl 1), S159–S172.
- Kinane, D. F., Stathopoulou, P. G., & Papapanou, P. N. (2017). Periodontal diseases. Nature Reviews Disease Primers, 3, 17038.
- Socransky, S. S., & Haffajee, A. D. (2005). Periodontal microbial ecology. Periodontology 2000, 38(1), 135–187.
- Bartold, P. M., & Van Dyke, T. E. (2013). Periodontitis: A host-mediated disruption of microbial homeostasis. Unlearning learned concepts. Periodontology 2000, 62(1), 203–217.
- Offenbacher, S. (1996). Periodontal diseases: Pathogenesis. Annals of Periodontology, 1(1), 821–878.
- Pihlstrom, B. L., Michalowicz, B. S., & Johnson, N. W. (2005). Periodontal diseases. The Lancet, 366(9499), 1809–1820.
- Slots, J. (2017). Periodontitis: Facts, fallacies and the future. Periodontology 2000, 75(1), 7–23.
- Chapple, I. L. C., Mealey, B. L., Van Dyke, T. E., Bartold, P. M., Dommisch, H., Eickholz, P., Geisinger, M. L., Genco, R. J., Glogauer, M., Goldstein, M., et al. (2018). Periodontal health and gingival diseases and conditions on an intact and a reduced periodontium: Consensus report of workgroup 1 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. Journal of Periodontology, 89(Suppl 1), S74–S84.
- Trombelli, L., Farina, R., Silva, C. O., & Tatakis, D. N. (2018). Plaque-induced gingivitis: Case definition and diagnostic considerations. Journal of Periodontology, 89(Suppl 1), S46–S73.
- Lang, N. P., & Bartold, P. M. (2018). Periodontal health. Journal of Periodontology, 89(Suppl 1), S9–S16.
- Cortellini, P., & Tonetti, M. S. (2015). Clinical concepts for regenerative therapy in intrabony defects. Periodontology 2000, 68(1), 282–307.
- Van Dyke, T. E., & Dave, S. (2005). Risk factors for periodontitis. Journal of the International Academy of Periodontology, 7(1), 3–7.
- American Academy of Periodontology (AAP). (2015). Comprehensive Periodontal Therapy: A Statement by the American Academy of Periodontology. Journal of Periodontology, 82(7), 943–949.
- Highfield, J. (2009). Diagnosis and classification of periodontal disease. Australian Dental Journal, 54(Suppl 1), S11–S26.
- Nanci, A., & Bosshardt, D. D. (2006). Structure of periodontal tissues in health and disease. Periodontology 2000, 40(1), 11–28.
- Meyle, J., & Chapple, I. (2015). Molecular aspects of the pathogenesis of periodontitis. Periodontology 2000, 69(1), 7–17.
- Slots, J., & Slots, H. (2019). Periodontitis: Facts, fallacies and the future. Periodontology 2000, 79(1), 7–23.