Biofilms are complex microbial communities that form on various surfaces, including teeth, medical devices, and natural environments. This article delves into the fascinating world of biofilm formation, exploring what biofilms are, how they develop, and the implications they have in different fields. Understanding the nature of biofilms is crucial for addressing the challenges they pose in healthcare settings, industrial processes, and environmental systems.
Table of Contents
ToggleWhat are Biofilms?
Biofilms are complex microbial communities that form on various surfaces, both natural and man-made. They consist of microorganisms, such as bacteria, fungi, and algae, embedded in a self-produced matrix of extracellular polymeric substances (EPS). This matrix, often referred to as the “sticky substance,” is a key characteristic of biofilms.
Defining Biofilms
Biofilms are defined as structured communities of microorganisms that adhere to surfaces and are surrounded by a protective extracellular matrix. This matrix is composed of a combination of polysaccharides, proteins, nucleic acids, and other organic and inorganic materials. The matrix acts as a glue, allowing the microorganisms to adhere to surfaces and form a cohesive community.
The Structure of Biofilms
Biofilms have a complex three-dimensional structure. They consist of microcolonies or clusters of microorganisms, which are embedded within the EPS matrix. This structure provides protection and allows for the exchange of nutrients and waste products within the biofilm.
Microorganisms Involved in Biofilm Formation
Biofilms can be formed by various microorganisms, including bacteria, fungi, algae, and protozoa. Bacterial biofilms are the most extensively studied and commonly encountered in different environments. Within a biofilm, different species of microorganisms can coexist, forming a diverse microbial community.
Understanding biofilms is crucial because they have significant implications in various fields, including healthcare, industry, and the environment. Biofilms play a role in medical device-associated infections, biofouling of industrial equipment, and ecological processes in natural ecosystems.
Stages of Biofilm Formation
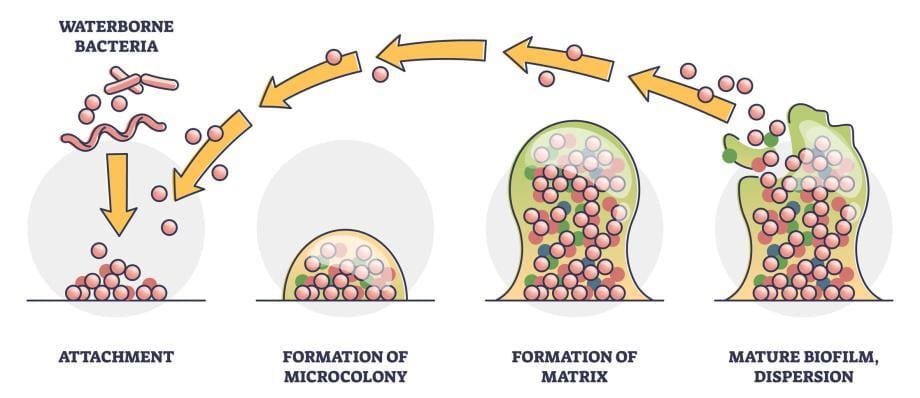
Biofilm formation typically occurs in a series of stages, each characterized by distinct processes and events. Understanding these stages is essential for comprehending the development and maturation of biofilms.
- Attachment Phase: Finding a Surface
- Microcolony Formation: Building the Foundation
- Biofilm Maturation: Developing a Complex Structure
- Dispersion: Spreading and Dissemination
Attachment Phase: Finding a Surface
The first stage of biofilm formation is the attachment phase. Microorganisms, often in a planktonic (free-floating) state, encounter a suitable surface to colonize. They utilize various mechanisms, such as pili, fimbriae, or adhesion proteins, to adhere to the surface. This initial attachment is reversible, allowing for exploration and potential detachment if the conditions are unfavorable.
Microcolony Formation: Building the Foundation
Once attached, microorganisms begin to multiply and form microcolonies on the surface. The microcolonies serve as the foundation for biofilm development. Within these microcolonies, microbial cells communicate through a process known as quorum sensing, which involves the exchange of chemical signals. Quorum sensing helps coordinate gene expression and behavior within the biofilm community.
Biofilm Maturation: Developing a Complex Structure
During the maturation stage, the biofilm undergoes significant structural development. Microorganisms continue to multiply and produce EPS, which contributes to the formation of a three-dimensional architecture. The EPS matrix not only provides structural stability but also acts as a protective barrier, shielding the microorganisms from environmental stressors, antimicrobial agents, and the host immune response.
Dispersion: Spreading and Dissemination
In certain circumstances, biofilms can enter a dispersal phase. This phase involves the detachment of cells or small clusters from the biofilm, allowing them to spread and potentially colonize new surfaces. Dispersal can occur in response to environmental cues or as a survival strategy when conditions become unfavorable within the biofilm.
These stages of biofilm formation highlight the dynamic and evolving nature of biofilms. The attachment phase sets the foundation for subsequent growth and development, leading to the maturation of a complex biofilm structure. Dispersion allows for the potential dissemination of biofilm cells, contributing to the colonization of new surfaces and the spread of biofilm-related issues.
Understanding the stages of biofilm formation is crucial for developing strategies to prevent and control biofilm-related problems. By targeting specific stages, such as the initial attachment or the quorum sensing process, it may be possible to disrupt biofilm formation and limit their detrimental effects.
Factors Influencing Biofilm Formation
Biofilm formation is influenced by various factors, including surface characteristics, environmental conditions, nutrient availability, and microbial interactions. Understanding these factors is crucial for comprehending the complex dynamics of biofilm development and for developing strategies to prevent and control their formation.
- Surface Characteristics and Substrates
- Environmental Factors: Temperature, pH, and Oxygen Availability
- Nutrient Availability and Quorum Sensing
- Role of Microbial Interactions
Surface Characteristics and Substrates
The characteristics of the surface to which microorganisms attach play a significant role in biofilm formation. Surface roughness, hydrophobicity, charge, and composition can impact the initial attachment and subsequent growth of biofilms. Different surfaces may promote or inhibit biofilm formation, making surface modification an area of interest for biofilm control.
Environmental Factors: Temperature, pH, and Oxygen Availability
Environmental conditions, such as temperature, pH, and oxygen availability, can influence biofilm formation. Microorganisms have specific temperature and pH ranges in which they thrive, and deviations from these optimal conditions can affect biofilm development. Oxygen availability also plays a role, as some microorganisms prefer aerobic conditions, while others thrive in anaerobic environments.
Nutrient Availability and Quorum Sensing
The availability of nutrients is crucial for biofilm formation. Biofilms require a source of carbon, nitrogen, and other essential elements for microbial growth and EPS production. Nutrient availability can influence the rate of biofilm formation and the overall biomass of the biofilm. Quorum sensing, a cell-to-cell communication mechanism, also plays a role in coordinating biofilm development and regulating gene expression within the microbial community.
Role of Microbial Interactions
Microbial interactions within biofilms can greatly influence their formation and development. Synergistic interactions, where microorganisms cooperate and benefit from each other’s activities, can enhance biofilm growth and stability. Conversely, competition between microorganisms for resources can limit biofilm formation. Antagonistic interactions, where one microorganism inhibits the growth of others through the production of antimicrobial substances, can also affect biofilm dynamics.
Understanding the factors that influence biofilm formation is crucial for developing effective strategies to prevent and control their growth. Surface modifications, environmental control, nutrient management, and targeted disruption of microbial interactions are all potential approaches to manage biofilms. By manipulating these factors, it may be possible to limit biofilm formation and mitigate the associated risks in various settings.
Biofilms in Healthcare Settings
Biofilms pose significant challenges in healthcare settings, as they can lead to persistent and recurrent infections. This section explores the impact of biofilms on healthcare-associated infections, the challenges in treating biofilm-related infections, and strategies to combat these issues.
- Biofilm-Associated Infections
- Challenges in Treating Biofilm Infections
- Strategies to Combat Biofilm-Related Infections
Biofilm-Associated Infections
Biofilms are commonly associated with infections related to medical devices, such as catheters, implants, and prosthetic devices. Once formed, biofilms provide a protective environment for microorganisms, making them highly resistant to antimicrobial agents and host immune responses. Biofilm-associated infections can lead to complications, prolonged hospital stays, increased healthcare costs, and even mortality.
Challenges in Treating Biofilm Infections
The inherent resistance of biofilms to antibiotics and disinfectants poses significant challenges in treating biofilm-related infections. The biofilm matrix acts as a physical barrier, preventing antimicrobial agents from reaching the microorganisms within the biofilm. Additionally, biofilm cells can enter a dormant state, further reducing their susceptibility to antimicrobial treatments.
Strategies to Combat Biofilm-Related Infections
Efforts are being made to develop strategies to prevent and control biofilm-related infections in healthcare settings. Some approaches include the development of antimicrobial coatings for medical devices, the use of antimicrobial agents specifically targeting biofilms, and the exploration of novel technologies such as photodynamic therapy and ultrasound to disrupt and kill biofilm cells. Improving the design of medical devices to discourage biofilm formation and promoting strict adherence to infection control practices are also crucial in preventing biofilm-associated infections.
By understanding the unique challenges posed by biofilms in healthcare settings, researchers and healthcare professionals can work together to develop effective strategies to prevent biofilm formation, enhance the treatment of biofilm-related infections, and improve patient outcomes.
Biofilms in Industrial Processes
Biofilms have significant implications in various industrial processes, ranging from food and beverage production to water treatment systems. This section explores the impact of biofilms on industrial equipment, the challenges they pose in the food and beverage industries, and their role in biofouling of water treatment systems.
- Biofilms and Industrial Equipment
- Implications for Food and Beverage Industries
- Biofouling in Water Treatment Systems
Biofilms and Industrial Equipment
Biofilms can form on industrial equipment surfaces, such as pipes, tanks, and heat exchangers. The presence of biofilms on these surfaces can lead to several issues. Biofilms can cause corrosion, decrease heat transfer efficiency, and clog pipes, leading to decreased productivity and increased maintenance costs. Additionally, biofilms can serve as a reservoir for pathogens, potentially contaminating the final products.
Implications for Food and Beverage Industries
Biofilms pose particular challenges in the food and beverage industries, where cleanliness and product safety are paramount. Biofilms can form on processing equipment, packaging materials, and storage areas. They can cause product contamination, affect the quality and shelf-life of food and beverages, and increase the risk of foodborne illnesses. Preventing and managing biofilms in these industries require rigorous cleaning and sanitation protocols, as well as the use of antimicrobial agents and regular equipment maintenance.
Biofouling in Water Treatment Systems
Biofilms can also cause biofouling in water treatment systems. When biofilms form on the surfaces of filters, membranes, and pipes, they can decrease water flow rates, reduce the efficiency of treatment processes, and promote the growth of pathogenic bacteria. Effective biofilm control strategies, such as routine cleaning, disinfection, and the use of specialized coatings, are essential to maintain the performance and longevity of water treatment systems.
Understanding the impact of biofilms in industrial processes is crucial for developing strategies to prevent and control their formation. Regular cleaning and maintenance protocols, surface modifications to discourage biofilm attachment, and the use of antimicrobial agents are some approaches employed in industrial settings to manage biofilms effectively.
Environmental Impact of Biofilms
Biofilms have significant implications in natural ecosystems, both aquatic and terrestrial. This section explores the role of biofilms in ecological processes, their impact on environmental health, and their potential applications in bioremediation.
- Role of Biofilms in Ecological Processes
- Biofilms and Environmental Health
- Bioremediation Applications
Role of Biofilms in Ecological Processes
Biofilms play a crucial role in various ecological processes. In aquatic environments, biofilms form on rocks, sediment, and other submerged surfaces. They serve as a habitat and a source of food for many organisms, including invertebrates and fish. Biofilms also contribute to nutrient cycling, oxygen production, and the breakdown of organic matter, playing a vital role in the overall functioning of aquatic ecosystems.
Biofilms and Environmental Health
While biofilms play essential roles in ecosystems, they can also have negative impacts on environmental health. Excessive biofilm growth can lead to the eutrophication of water bodies, where an overabundance of nutrients promotes the proliferation of harmful algal blooms and depletes oxygen levels. Biofilms can also act as reservoirs for pollutants and pathogens, contributing to water and soil contamination.
Bioremediation Applications
Biofilms have shown potential in environmental remediation efforts. Certain microorganisms within biofilms have the ability to degrade pollutants, such as hydrocarbons, heavy metals, and pesticides. Biofilm-based bioremediation strategies can be employed to clean up contaminated soils, sediments, and wastewater. These strategies harness the metabolic capabilities of biofilm-associated microorganisms to break down and remove pollutants from the environment.
Understanding the environmental impact of biofilms is crucial for maintaining ecological balance and addressing pollution issues. By studying the interactions between biofilms and the environment, scientists can develop strategies to mitigate the negative effects of biofilms on environmental health and harness their potential for bioremediation purposes.
Future Directions and Research Prospects
Biofilm research is a rapidly evolving field, and ongoing studies continue to deepen our understanding of biofilm formation, behavior, and management. This section explores potential future directions and research prospects in biofilm studies.
- Biofilm Prevention and Control Strategies
- Biofilm-Host Interactions
- Biofilms in Novel Environments
- Multi-Disciplinary Approaches
- Biofilms and Human Health
Biofilm Prevention and Control Strategies
One area of future research in biofilm studies revolves around the development of novel prevention and control strategies. Researchers are investigating innovative approaches to disrupt biofilm formation, such as the use of antimicrobial peptides, nanoparticles, and biofilm-targeting enzymes. Advancements in surface engineering techniques, including the development of self-cleaning surfaces and coatings resistant to biofilm attachment, are also promising avenues for biofilm management.
Biofilm-Host Interactions
Understanding the intricate interactions between biofilms and the host is another area of research interest. Scientists are investigating the host immune response to biofilms, including the role of immune cells and the potential modulation of immune responses to better combat biofilm-associated infections. Furthermore, exploring the communication and signaling between biofilms and host cells can provide insights into the pathogenesis of biofilm-related diseases.
Biofilms in Novel Environments
As our understanding of biofilms expands, researchers are exploring biofilm formation and behavior in novel environments. This includes studying biofilms in extreme environments, such as deep-sea hydrothermal vents and polar regions, as well as biofilms associated with emerging technologies and materials. Investigating biofilms in these unique settings can shed light on their adaptability and potential applications in various industries.
Multi-Disciplinary Approaches
Biofilm research benefits from a multi-disciplinary approach, and future studies are expected to continue integrating diverse fields such as microbiology, engineering, materials science, and computational modeling. Collaborative efforts between researchers with different expertise can lead to innovative solutions for biofilm prevention, control, and treatment.
Biofilms and Human Health
Further understanding the implications of biofilms on human health is also a promising research direction. Investigating the role of biofilms in chronic infections, such as periodontitis and cystic fibrosis, can provide insights into disease progression and potential therapeutic interventions. Additionally, exploring the impact of biofilms on the microbiome and their interactions with the human immune system can advance our knowledge of host-microbe dynamics.
Overall, biofilm research is a dynamic and promising field with a wide range of research prospects, and further exploration in these areas can lead to breakthroughs in biofilm prevention, control, and treatment.
Conclusion
Biofilm formation is a natural phenomenon that has significant implications in various fields. Understanding the stages and factors influencing biofilm development is crucial for developing effective strategies to manage biofilm-related challenges. In healthcare settings, combating biofilm-associated infections remains a major concern, while in industrial processes, biofilms can lead to equipment damage and product contamination. Environmental systems also experience the impacts of biofilms, affecting water treatment and ecological balance. Continued research and the development of innovative approaches are essential to overcome the challenges posed by biofilm formation and maintain a clean and healthy environment in different settings