Periodontal disease often results in the destruction of tooth-supporting structures such as the gingiva, periodontal ligament, cementum, and alveolar bone. The primary goal of periodontal therapy is to halt the progression of the disease and regenerate the lost tissues, thereby restoring normal form and function. Periodontal regeneration surgery encompasses a range of advanced techniques designed to promote the reformation of these lost tissues, rather than merely repairing them.
Among these, Guided Tissue Regeneration (GTR), bone grafting, and enamel matrix derivatives (EMDs) represent the cornerstone approaches in modern periodontal regeneration.
Table of Contents
ToggleBiological Basis of Periodontal Regeneration
True periodontal regeneration refers to the formation of new cementum, new periodontal ligament fibers inserted into the new cementum, and new alveolar bone on a previously diseased root surface. This differs from repair, which results only in the re-establishment of tissue continuity without restoring the original architecture or function (e.g., formation of long junctional epithelium).
Histologically, true regeneration can only be confirmed by examining extracted specimens; however, in clinical practice, radiographic bone fill and probing depth reduction serve as surrogate measures of regenerative success.
The biological rationale for regenerative surgery originates from the understanding of wound healing following periodontal surgery. When a periodontal defect is treated surgically, several cell types compete to repopulate the root surface: epithelial cells, gingival connective tissue cells, bone cells, and periodontal ligament (PDL) cells. Studies have demonstrated that if epithelial cells migrate rapidly along the root surface, they prevent the attachment of PDL cells, leading to the formation of a long junctional epithelium instead of true regeneration. Hence, controlling epithelial migration became a central concept in regenerative therapy.
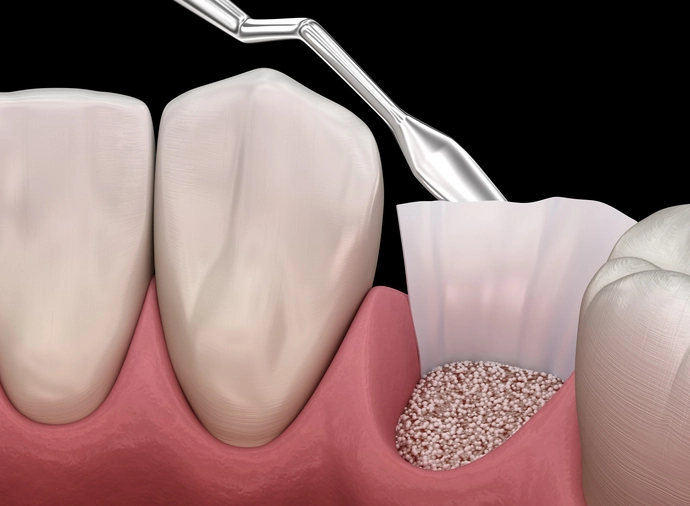
Guided Tissue Regeneration (GTR)
Historical Background
The concept of GTR emerged from the recognition that preventing epithelial migration along the root surface allows cells from the PDL and bone to repopulate the area, enabling new attachment formation. In 1982, Nyman and colleagues demonstrated this principle by placing a barrier membrane over an intrabony defect in a tooth planned for extraction. Histological examination revealed new cementum, new bone, and new PDL fibers, confirming true regeneration.
Principle and Mechanism
GTR is based on the concept of cellular exclusion — selectively allowing the proliferation of cells from the periodontal ligament and alveolar bone while excluding epithelial and gingival connective tissue cells. This is achieved through the placement of a barrier membrane between the gingival flap and the defect area.
The membrane maintains space for the slower-migrating PDL and bone cells to repopulate the area while preventing faster epithelial downgrowth. Over time, this leads to the formation of new connective tissue attachment and bone.
Barrier Membranes
Membranes used in GTR can be broadly classified as:
1. Non-resorbable membranes
- Early materials such as expanded polytetrafluoroethylene (ePTFE) were non-resorbable and required a second surgical procedure for removal.
- While effective, they posed challenges such as risk of infection or membrane exposure.
2. Resorbable membranes
- To overcome the drawbacks of non-resorbable materials, bioresorbable membranes were developed.
- Examples include Bio-Gide®, a collagen-based resorbable membrane, and Atrisorb® FreeFlow, a bioabsorbable polymer.
- These materials gradually degrade within the body, eliminating the need for a second surgery.
Procedure
- Flap reflection and defect debridement: The surgical site is accessed, and all granulation tissue is removed.
- Root surface preparation: Scaling and root planing are performed to create a clean, smooth surface.
- Membrane placement: The barrier membrane is positioned to cover the defect and stabilized to maintain space for regeneration.
- Flap repositioning and suturing: The flaps are carefully repositioned and sutured to ensure primary closure and stability.
- Postoperative care: The surgical site must remain undisturbed for several weeks to allow for tissue regeneration beneath the membrane.
Bone Grafts
Rationale
Bone grafting is another major regenerative technique used to restore alveolar bone lost due to periodontal disease. Bone grafts can be used alone or in combination with GTR membranes to enhance regenerative outcomes. The graft material serves as a scaffold (osteoconductive), provides inductive signals (osteoinductive), or supplies vital cells (osteogenic) that promote new bone formation.
Types of Bone Grafts
1. Autografts:
- Bone harvested from the patient’s own body (e.g., mandibular symphysis, ramus, or iliac crest).
- Considered the “gold standard” due to their osteogenic potential.
- Limitations include donor site morbidity and limited quantity.
2. Allografts:
- Bone obtained from another human donor, processed to remove antigenicity.
- Examples include demineralized freeze-dried bone allograft (DFDBA), which exhibits osteoinductive potential due to the presence of bone morphogenetic proteins (BMPs).
3. Xenografts:
- Derived from non-human species, typically bovine bone (e.g., Bio-Oss®).
- Primarily osteoconductive and widely used due to biocompatibility and structural similarity to human bone.
4. Alloplasts (synthetic bone substitutes):
- Synthetic materials such as bioactive glass (e.g., PerioGlas®) or hydroxyapatite.
- Serve as osteoconductive scaffolds that are gradually replaced by new bone.
Combination Therapy
GTR combined with bone grafts often yields superior results by promoting both soft tissue and bone regeneration. The membrane prevents epithelial ingrowth, while the graft material stabilizes the clot and provides a scaffold for new bone formation.
Enamel Matrix Derivatives (EMDs)
Background and Composition
Enamel matrix derivatives are biologically active proteins derived from developing porcine enamel matrix. Emdogain® is the most widely used commercial formulation, consisting of enamel matrix proteins suspended in a propylene glycol alginate gel.
These proteins, primarily amelogenins, mimic the natural events of root development by stimulating cementoblasts and PDL fibroblasts to produce new acellular cementum and collagen fibers.
Mechanism of Action
During tooth development, Hertwig’s epithelial root sheath (HERS) plays a key role in inducing the differentiation of cementoblasts and the formation of the cementum-PDL complex. EMDs replicate this biological environment, promoting:
- Cementogenesis
- Periodontal ligament formation
- Alveolar bone regeneration
The locally applied EMD solution on a prepared root surface initiates a cascade of biological events that resemble the natural process of periodontal tissue development.
Application Technique
- Flap reflection and root surface debridement: The root surface is thoroughly cleaned and conditioned with ethylene diamine tetraacetic acid (EDTA) to remove the smear layer.
- EMD application: EMD solution is applied to the root surface immediately after conditioning.
- Flap closure: The flap is repositioned and sutured to achieve a tension-free closure.
Indications for Regenerative Procedures
Successful regeneration depends largely on appropriate case selection and defect morphology.
- Intrabony defects:
Best results are achieved in two- or three-walled vertical defects.
Three-walled defects, being self-contained, have the highest regenerative potential. - Furcation defects:
Grade I furcation defects respond favorably to regenerative therapy.
Grade II defects have variable outcomes, while grade III defects show poor predictability. - Defect shape and size:
Deep and narrow defects show better regeneration than wide and shallow ones due to improved clot stability and space maintenance. - Horizontal bone loss:
Largely unpredictable and generally unsuitable for regenerative procedures.
Other factors affecting success include patient oral hygiene, smoking status, systemic health, and surgical technique. Smoking, in particular, has been consistently associated with reduced regenerative outcomes due to impaired wound healing and vascularity.
Surgical Technique (General Steps)
- Pre-surgical preparation: Thorough scaling, root planing, and infection control. Patient motivation and plaque control must be established.
- Flap design and access: A full-thickness mucoperiosteal flap is elevated to provide access to the defect.
- Debridement and root surface treatment: All granulation tissue is removed. Root surfaces are mechanically cleaned and chemically conditioned with EDTA if EMD is to be applied.
- Placement of regenerative material: Depending on the case, a bone graft, GTR membrane, or EMD is placed over the defect.
- Flap repositioning and suturing: Primary closure without tension is essential to protect the regenerative site.
- Postoperative care: Antibiotics, chlorhexidine rinses, and avoidance of trauma to the surgical area are recommended. Sutures are typically removed after 7–10 days.
Clinical Outcomes and Predictability
Guided Tissue Regeneration
Numerous clinical studies and systematic reviews have shown that GTR leads to significant gains in clinical attachment levels and bone fill in intrabony defects compared with conventional flap surgery. However, outcomes vary depending on defect morphology, patient factors, and surgical precision.
Bone Grafting
Bone grafts provide an osteoconductive framework and, in some cases, osteoinductive potential. When combined with GTR, grafts enhance defect fill and improve stability of the regenerative site.
Enamel Matrix Derivatives
A Cochrane review (Esposito et al., 2004) concluded that EMDs are effective in promoting periodontal regeneration in intrabony defects. Studies have demonstrated new formation of cementum, PDL, and alveolar bone. However, predictability remains limited and highly technique-sensitive.
Factors Influencing Success
- Defect morphology: Three-walled defects respond best.
- Flap stability: Primary closure and minimal micromotion are critical.
- Patient factors: Smoking, diabetes, and poor oral hygiene negatively affect outcomes.
- Material handling: Proper membrane adaptation and graft stability enhance results.
- Operator skill: Precision in surgical execution determines success.
Limitations and Challenges
While regenerative techniques have advanced significantly, several limitations persist:
- Difficulty in achieving complete regeneration in all cases.
- Variability in clinical outcomes due to patient-related factors.
- High technique sensitivity and cost.
- Potential for membrane exposure and infection in GTR.
- Limited predictability in horizontal defects and advanced furcation involvement.
Future Directions
Recent advancements in tissue engineering and biomaterials science hold promise for overcoming the limitations of current regenerative techniques. Research focuses on combining scaffolds, growth factors, and stem cells to promote more predictable regeneration.
Potential developments include:
- Use of growth factors such as platelet-derived growth factor (PDGF) and bone morphogenetic proteins (BMPs).
- 3D-printed scaffolds designed to fit defect morphology precisely.
- Gene therapy to enhance cellular regenerative capacity.
- Stem cell-based therapy using periodontal ligament stem cells (PDLSCs) to regenerate lost tissues.
These emerging approaches aim to achieve consistent, functional regeneration of cementum, PDL, and alveolar bone, and may eventually lead to complete restoration of lost periodontal structures.
Outcome and Prognosis
The regeneration of cementum, periodontal ligament, and alveolar bone is biologically possible and has been demonstrated histologically. However, clinical predictability remains moderate, and long-term stability depends on maintaining excellent oral hygiene and control of etiologic factors.
A systematic review (Sharpe et al., 2008) confirmed that regenerative procedures, when properly indicated and performed, yield superior outcomes compared to conventional periodontal therapy. Nevertheless, the variability in patient response underscores the need for careful case selection and meticulous surgical technique.
Conclusion
Regenerative periodontal surgery represents one of the most biologically and clinically significant advancements in modern periodontology. Techniques such as guided tissue regeneration, bone grafting, and enamel matrix derivatives aim to restore lost periodontal tissues to their original structure and function.
While true regeneration—defined as the new formation of cementum, PDL, and alveolar bone—has been demonstrated in controlled studies, clinical predictability remains influenced by defect morphology, patient factors, and surgical precision.
Future developments in biomaterials and tissue engineering are likely to revolutionize regenerative periodontics, bringing dentistry closer to achieving consistent, functional regeneration of the entire periodontium.
References
- Nyman S, Lindhe J, Karring T, Rylander H. (1982). New attachment following surgical treatment of human periodontal disease. J Clin Periodontol, 9:290–296.
- Sharpe G, et al. (2008). Dental Update, 35:304.
- Esposito M, et al. (2004). J Dent Educ, 68:834.
- Slavkin HC, Bartold PM. (2006). Periodontology 2000, 41:9.
- Lang NP, Tonetti MS. (2004). Periodontology 2000, 34:1–3.
- Sculean A, et al. (2015). J Clin Periodontol, 42:S6–S16.