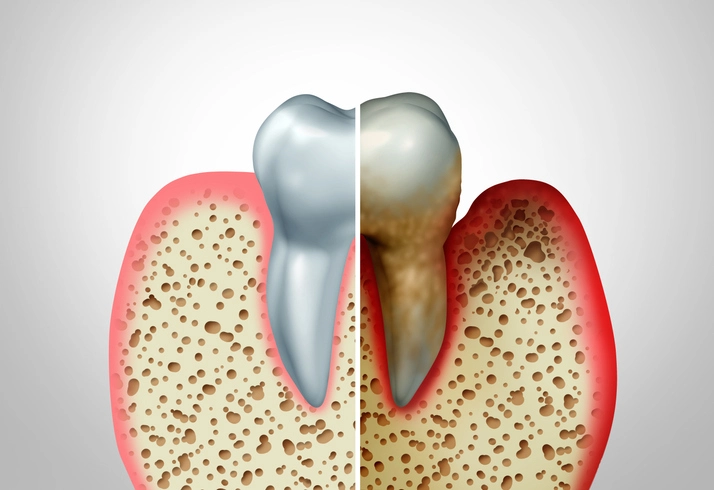
Periodontal diseases encompass a wide range of inflammatory conditions affecting the supporting structures of the teeth, including the gingiva, periodontal ligament, cementum, and alveolar bone. Among these, aggressive periodontitis and necrotizing periodontal diseases represent severe and rapidly progressive forms of periodontal pathology that demand urgent diagnosis and management.
Historically, aggressive periodontitis was considered a distinct clinical entity, characterized by rapid attachment loss and bone destruction in otherwise systemically healthy individuals. However, with advances in our understanding of host-pathogen interactions and the development of the 2017 World Workshop Classification of Periodontal and Peri-Implant Diseases, the distinction between “aggressive” and “chronic” periodontitis was removed. Instead, periodontitis is now classified based on its stage and grade, which reflect severity, extent, and rate of progression. Despite this change in terminology, the concept of rapidly progressive or “aggressive” disease remains clinically relevant.
Similarly, necrotizing periodontal diseases (NPDs) represent an acute, destructive spectrum of infections involving the gingiva and supporting periodontal structures. They are often associated with systemic or local predisposing factors such as immunosuppression, malnutrition, or extreme stress.
Table of Contents
ToggleAggressive Periodontitis
Historical Background and Classification
Aggressive periodontitis (AgP) was recognized as a unique form of periodontal disease due to its rapid progression and occurrence in young, systemically healthy individuals. The 1999 classification system defined it as a distinct clinical category, separate from chronic periodontitis. However, the 2017 Classification of Periodontal Diseases reclassified it as a high-grade periodontitis, acknowledging that both chronic and aggressive forms share similar microbial etiologies and host responses, differing mainly in their rate of progression and age of onset.
Aggressive periodontitis was traditionally divided into two major subtypes:
- Localized Aggressive Periodontitis (LAP)
- Generalized Aggressive Periodontitis (GAP)
These subtypes exhibit distinct patterns of clinical presentation, microbial colonization, and immune responses.
Epidemiology
The prevalence of aggressive periodontitis varies globally, with higher incidences reported in individuals of African descent compared to Caucasian or Asian populations. Localized aggressive periodontitis affects approximately 0.1% to 2% of the population, whereas generalized forms are less common. The disease typically manifests during adolescence or early adulthood, with a familial pattern suggesting a genetic predisposition.
Etiopathogenesis
Aggressive periodontitis is a multifactorial disease resulting from a complex interplay between bacterial infection and host immune response.
1. Microbial Factors
The primary pathogen associated with AgP is Aggregatibacter actinomycetemcomitans (A.a), a Gram-negative facultative anaerobe known for its ability to invade host tissues and produce virulence factors such as leukotoxin, endotoxin, and proteases.
Other microorganisms often implicated include Porphyromonas gingivalis, Prevotella intermedia, and Capnocytophaga species.
2. Host Response
Individuals with aggressive periodontitis exhibit an exaggerated immune-inflammatory response. Neutrophil chemotaxis and phagocytosis are often impaired, leading to inadequate bacterial clearance. Additionally, macrophages and monocytes in affected patients may produce higher levels of pro-inflammatory cytokines such as IL-1β and TNF-α, accelerating tissue destruction.
3. Genetic Predisposition
Familial aggregation is well-documented in AgP, suggesting a strong genetic component. Polymorphisms in genes encoding interleukin-1 (IL-1), Fcγ receptors, and cathepsin C have been associated with increased susceptibility.
4. Environmental and Behavioral Factors
Smoking, poor oral hygiene, and psychosocial stress are recognized as major risk modifiers that exacerbate disease progression.
Clinical Features
1. Localized Aggressive Periodontitis (LAP)
- Age of onset: Around puberty.
- Pattern: Localized attachment and bone loss around first molars and incisors.
- Extent: Involves at least two permanent teeth (one being a first molar) and no more than two other teeth.
- Plaque levels: Often minimal, inconsistent with the severity of destruction.
- Inflammation: Gingival tissues may appear clinically normal despite deep pockets and bone loss.
- Serum antibody response: Robust antibody response against infecting agents, especially A. actinomycetemcomitans.
2. Generalized Aggressive Periodontitis (GAP)
- Age of onset: Usually under 30 years but may appear later.
- Pattern: Generalized interproximal attachment loss affecting at least three permanent teeth other than first molars and incisors.
- Course: Episodic bursts of rapid destruction followed by periods of quiescence.
- Inflammation: Pronounced gingival inflammation with heavy plaque and calculus deposits during active phases.
- Antibody response: Poor serum antibody response compared to LAP.
Diagnosis
Diagnosis is based on:
- Clinical examination revealing disproportionate attachment loss relative to plaque accumulation.
- Radiographic evidence of angular bone defects.
- Family history suggesting hereditary susceptibility.
- Microbiological testing for A. actinomycetemcomitans or other specific pathogens (if available).
- Immune function tests may reveal defective neutrophil function.
Treatment and Management
The management of aggressive periodontitis requires a combination of mechanical therapy, antimicrobial therapy, and supportive maintenance.
1. Initial Phase: Infection Control
- Supragingival and subgingival scaling and root planing (SRP) to disrupt bacterial biofilms.
- Oral hygiene instruction and plaque control are essential to prevent reinfection.
2. Adjunctive Antimicrobial Therapy
Because of the invasive nature of the pathogens, systemic antibiotics are often necessary in addition to mechanical therapy.
The preferred regimen includes:
Amoxicillin (500 mg) + Metronidazole (400 mg), three times daily for 7–10 days.
This combination effectively targets A. actinomycetemcomitans and P. gingivalis.
For patients allergic to penicillin, azithromycin (500 mg once daily for 3 days) may be prescribed.
3. Surgical Intervention
Surgical therapy may be indicated for residual pockets or osseous defects not responding to non-surgical therapy. Options include:
- Flap surgery for access and debridement.
- Regenerative procedures (guided tissue regeneration, bone grafts) in localized defects.
Evidence supports the use of regenerative surgery in appropriate cases to restore lost attachment.
4. Supportive Periodontal Therapy
Long-term maintenance is crucial, involving:
- 3-month recall intervals for professional cleaning.
- Monitoring of pocket depths and bone levels.
- Reinforcement of oral hygiene practices.
Failure to maintain supportive care is the most common cause of recurrence.
Necrotizing Periodontal Diseases (NPDs)
Overview
Necrotizing periodontal diseases are acute, painful, and destructive infections of the gingival and periodontal tissues. They include:
- Necrotizing Ulcerative Gingivitis (NUG)
- Necrotizing Ulcerative Periodontitis (NUP)
- Necrotizing Stomatitis
These conditions are characterized by ulceration and necrosis of the gingival margins and interdental papillae, often associated with systemic factors such as malnutrition, immunodeficiency (especially HIV infection), stress, and smoking.
Etiology and Predisposing Factors
1. Microbial Factors
The microbiota associated with NPDs is distinctive and dominated by fusiform and spirochete organisms, including:
- Fusobacterium nucleatum
- Prevotella intermedia
- Treponema spp.
- Selenomonas spp.
These anaerobic bacteria invade the gingival epithelium and connective tissue, causing widespread necrosis.
2. Host and Systemic Factors
- Immunosuppression: HIV/AIDS, malnutrition, or chemotherapy.
- Psychological stress: Alters cortisol levels and impairs immune function.
- Smoking: Reduces oxygen tension in gingival tissues, favoring anaerobic growth.
- Poor oral hygiene: Allows accumulation of pathogenic biofilm.
Clinical Features
Necrotizing Ulcerative Gingivitis (NUG)
- Sudden onset of painful, bleeding gums.
- Punched-out, crater-like lesions at the interdental papillae covered with a grayish pseudomembrane.
- Fetid odor, metallic taste, and excessive salivation.
- Regional lymphadenopathy, malaise, and fever may be present.
Necrotizing Ulcerative Periodontitis (NUP)
- Represents progression of NUG into deeper periodontal structures, causing attachment loss and bone destruction.
- Patients may experience tooth mobility and localized necrosis of alveolar bone.
Necrotizing Stomatitis
- The most severe form, extending beyond mucogingival junction to involve soft tissue and bone.
- In extreme malnutrition or immune suppression, it may progress to noma (cancrum oris), a gangrenous condition of the face and jaws.
Differential Diagnosis
NPDs must be differentiated from:
- Primary herpetic gingivostomatitis.
- Desquamative gingivitis.
- Acute leukemia or agranulocytosis.
- Other vesiculobullous diseases.
Clinical history, hematological evaluation, and viral testing may aid in distinguishing these entities.
Microbiology
The characteristic microbiological pattern of NPDs involves a fusospirochetal complex. Treponema, Prevotella intermedia, and Fusobacterium species play central roles. These bacteria exhibit synergistic pathogenicity, invading necrotic tissues and releasing proteolytic enzymes, collagenases, and endotoxins that destroy connective tissue and bone.
Histologically, NUG lesions display four distinct zones:
- Bacterial zone – surface layer of spirochetes and fusiform bacteria.
- Neutrophil-rich zone – leukocytic infiltration with bacteria.
- Necrotic zone – disintegrated cells and tissue debris.
- Zone of spirochetal infiltration – viable tissue invaded by spirochetes.
Management
1. Acute Phase Management
The goal of initial therapy is pain relief, infection control, and tissue debridement.
Gentle debridement under local anesthesia to remove necrotic tissue and plaque.
Chlorhexidine mouth rinse (0.12–0.2%) twice daily.
Systemic antibiotics (if systemic signs present):
Metronidazole 200–400 mg, three times daily for 3 days, or
Penicillin V 250 mg four times daily for 7 days.
Analgesics for pain relief.
Rehydration and nutrition support for debilitated patients.
2. Subacute Phase
Once pain and acute inflammation subside (within 48–72 hours), scaling and root planing can be performed to remove residual calculus. Oral hygiene instruction should be reinforced.
3. Maintenance Phase
Regular supportive therapy to prevent recurrence is essential. Underlying systemic or behavioral factors must be addressed:
- Smoking cessation programs.
- Stress management and improved nutrition.
- Screening for HIV or systemic illnesses.
Complications
If untreated, necrotizing periodontal diseases can progress to extensive tissue necrosis, bone sequestration, orofacial deformity, and systemic sepsis. In immunocompromised individuals, recurrence is common, and the disease may become chronic.
Prognosis
The prognosis for both aggressive and necrotizing periodontal diseases depends on:
- Early diagnosis and prompt treatment.
- Effective infection control and patient compliance.
- Elimination of risk factors such as smoking or immune suppression.
- Regular maintenance therapy.
With proper management, long-term stability and function can be achieved, although some degree of attachment and bone loss may be irreversible.
Conclusion
Aggressive and necrotizing periodontal diseases, though uncommon, represent critical conditions that challenge both the clinician and the patient. Understanding their distinct clinical presentations, microbial profiles, and host interactions is essential for accurate diagnosis and effective management.
The shift in modern periodontal classification emphasizes disease progression rate and host susceptibility rather than rigid categories, highlighting that periodontitis is a spectrum of manifestations of a common underlying pathology.
Successful outcomes depend on a multidisciplinary approach — integrating mechanical debridement, antimicrobial therapy, patient education, and long-term maintenance. Moreover, clinicians should always consider systemic conditions like HIV infection, diabetes, or genetic predisposition when managing these aggressive diseases.
Ultimately, the cornerstone of periodontal therapy remains early detection, consistent maintenance, and personalized care aimed at preserving both oral health and overall well-being.
References
- Armitage GC. (1999). Development of a classification system for periodontal diseases and conditions. Annals of Periodontology, 4(1):1–6.
- Caton JG, Armitage GC, Berglundh T, et al. (2018). A new classification scheme for periodontal and peri‐implant diseases and conditions – Introduction and key changes from the 1999 classification. Journal of Clinical Periodontology, 45(Suppl 20):S1–S8.
- Fine DH, Patil AG, Loos BG. (2018). Classification and diagnosis of aggressive periodontitis. Journal of Periodontology, 89(Suppl 1):S103–S119.
- Lang NP, Bartold PM. (2018). Periodontal health. Journal of Clinical Periodontology, 45(Suppl 20):S9–S16.
- Slots J, Ting M. (1999). Actinobacillus actinomycetemcomitans and Porphyromonas gingivalis in human periodontitis: occurrence and treatment. Periodontology 2000, 20(1):82–121.
- Albandar JM, Tinoco EMB. (2002). Global epidemiology of aggressive periodontitis. Periodontology 2000, 29(1):7–13.
- Van Winkelhoff AJ, Rodenburg JP, Goené RJ, Abbas F, Winkel EG, de Graaff J. (1989). Metronidazole plus amoxicillin in the treatment of Actinobacillus actinomycetemcomitans associated periodontitis. Journal of Clinical Periodontology, 16(2):128–131.
- Tonetti MS, Greenwell H, Kornman KS. (2018). Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. Journal of Periodontology, 89(Suppl 1):S159–S172.
- Holmstrup P, Westergaard J. (2018). Necrotizing periodontal diseases: pathogenesis, clinical features, and management. Periodontology 2000, 76(1):26–49.
- Herrera D, Retamal-Valdes B, Alonso B, Feres M. (2018). Acute periodontal lesions (periodontal abscesses and necrotizing periodontal diseases) and endo-periodontal lesions. Journal of Clinical Periodontology, 45(Suppl 20):S78–S94.
- Pihlstrom BL, Michalowicz BS, Johnson NW. (2005). Periodontal diseases. The Lancet, 366(9499):1809–1820.
- Lindhe J, Lang NP, Karring T. (2008). Clinical Periodontology and Implant Dentistry, 5th Edition. Wiley-Blackwell.
- Carranza FA, Newman MG, Takei HH, Klokkevold PR. (2015). Carranza’s Clinical Periodontology, 12th Edition. Elsevier Saunders.
- Nunn M. (2003). Understanding the etiology of periodontitis: A historical perspective. Periodontology 2000, 32(1):11–23.
- Mealey BL, Ocampo GL. (2007). Diabetes mellitus and periodontal disease. Periodontology 2000, 44(1):127–153.
- Seymour GJ, Ford PJ, Cullinan MP, Leishman S, Yamazaki K. (2007). Relationship between periodontal infections and systemic disease. Clinical Microbiology and Infection, 13(Suppl 4):3–10.
- Williams RC, Mahan CJ. (1968). Periodontal diseases: the emergence of a new paradigm. Annals of Periodontology, 3(1):1–6.